Login
Welcome back! Please enter your details.
or
Don't have an account? Register here
Create Account
Join MedMentorEdu and start your medical journey.
or
Already have an account? Login here
Enhance your knowledge with our comprehensive guide and curated study materials.
Some Physico-chemical Laws Applied to Physiology (Biophysics)

• Biophysics deals with application of physical and chemical laws to biological systems.
• Many physiological processes follow physico-chemical principles.
• Important processes include:
• Diffusion
• Filtration
• Osmosis
• Electrolyte balance.
• These laws explain movement of substances across biological membranes.



• Concentration expresses amount of solute dissolved in a solvent.
• Important units used in physiology include:
• Molarity – number of moles of solute per litre of solution.
• Normality – number of gram equivalents per litre of solution.
• Percentage concentration – grams of solute in 100 mL of solution.
• Osmolarity – concentration of osmotically active particles.
• These units are useful in physiological and clinical calculations.


• Substances in solution may exist as ions or molecules.
• Based on their dissociation in water they are classified as:
• Ions
• Electrolytes
• Non-electrolytes.
• Ions are electrically charged particles formed by dissociation of substances in solution.
• Positive ions (cations) – sodium (Na⁺), potassium (K⁺), calcium (Ca²⁺).
• Negative ions (anions) – chloride (Cl⁻), bicarbonate (HCO₃⁻).
• Ions are essential for nerve conduction, muscle contraction and fluid balance.
• Electrolytes are substances that dissociate into ions in aqueous solution.
• They conduct electric current when dissolved in water.
• Examples include sodium chloride, potassium chloride and calcium salts.
• Electrolytes are important for osmotic balance and physiological functions.
• Non-electrolytes do not dissociate into ions in solution.
• They remain as uncharged molecules.
• Examples include glucose, urea and alcohol.
• They do not conduct electric current in solution.


• Filtration is movement of fluid and dissolved substances through a membrane due to pressure difference.
• Driven mainly by hydrostatic pressure.
• Occurs across capillary walls and kidney glomerulus.
• Important in formation of tissue fluid from capillaries.
• Essential in glomerular filtration in kidneys.
• Helps remove waste products from blood.
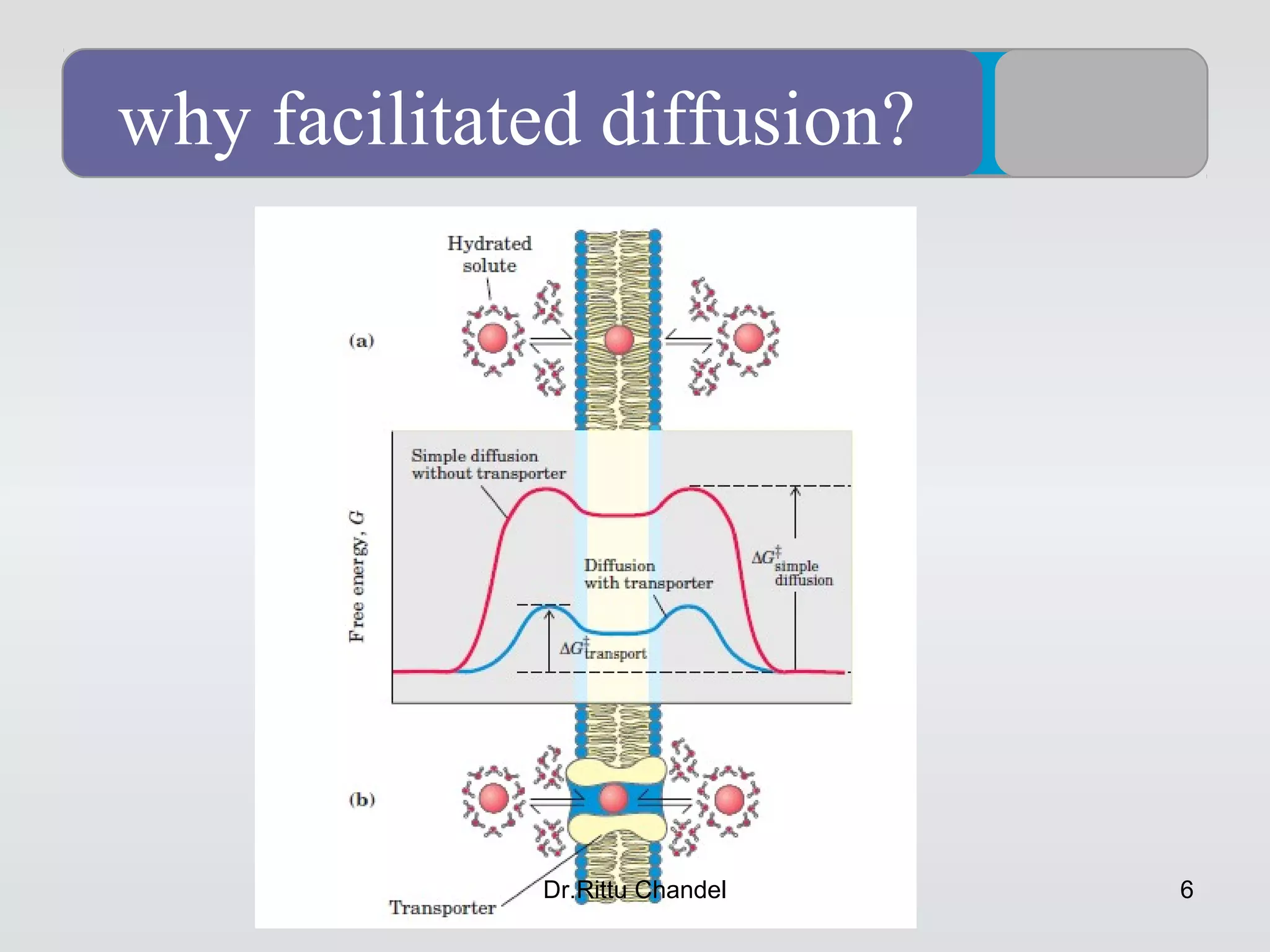
• Diffusion is movement of molecules from higher concentration to lower concentration.
• Occurs due to random molecular motion.
• Does not require energy expenditure.
• Continues until equilibrium is reached.
• Examples in physiology:
• Exchange of oxygen and carbon dioxide in lungs.
• Movement of solutes across cell membranes.


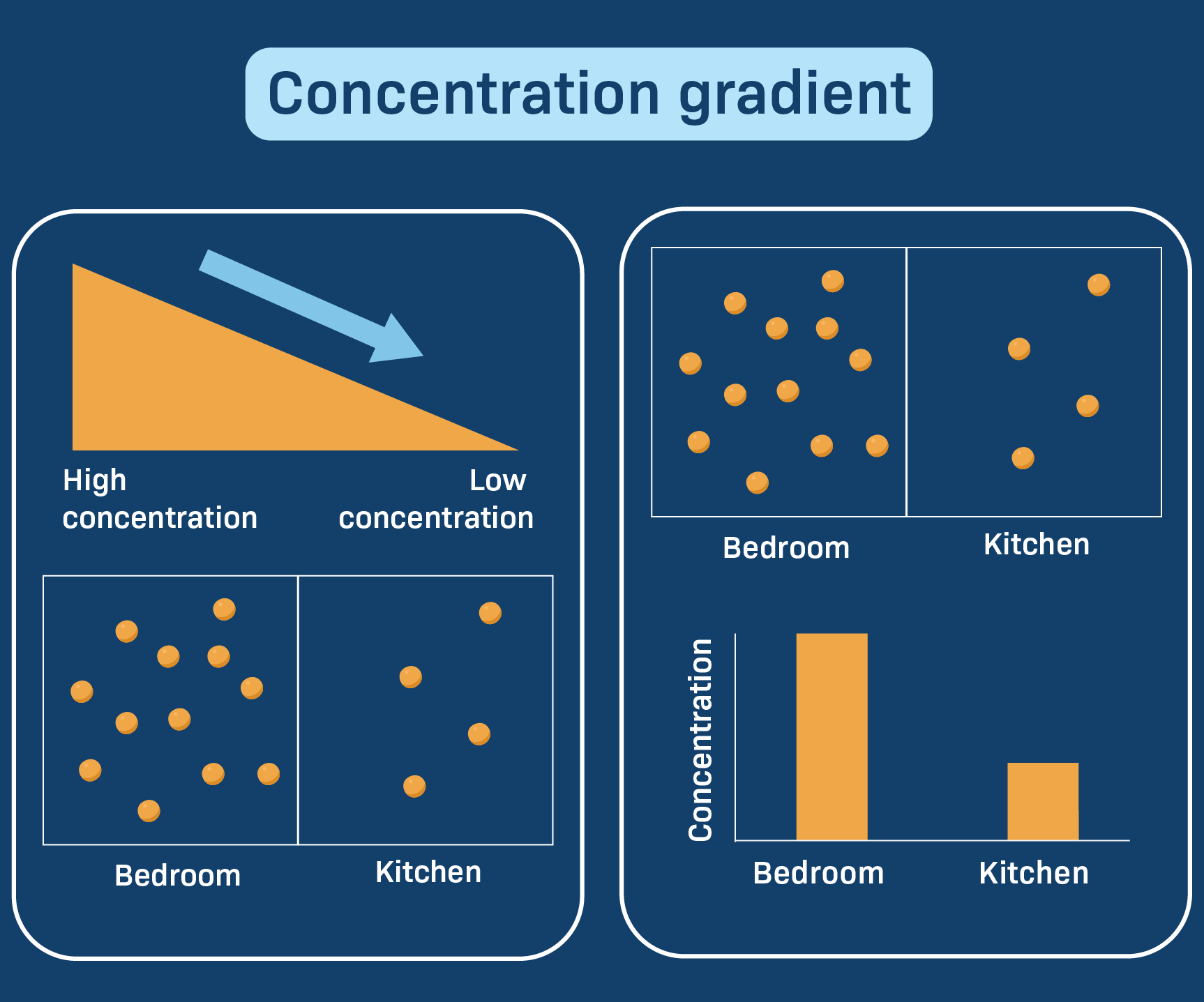
• Concentration refers to the amount of solute present in a given volume of solution.
• A concentration gradient exists when two regions have different solute concentrations.
• Diffusion occurs along this gradient from higher to lower concentration.
• Concentration gradient is a major driving force for diffusion in physiological processes.
Biology often looks complicated, but at its core it obeys the same simple rules as chemistry and physics: particles drift from crowded places to emptier ones, pressure pushes fluids through filters, and electric charges guide ions—life simply organizes these basic forces into astonishingly complex systems.
Get the full PDF version of this chapter.