Login
Welcome back! Please enter your details.
or
Don't have an account? Register here
Create Account
Join MedMentorEdu and start your medical journey.
or
Already have an account? Login here
Enhance your knowledge with our comprehensive guide and curated study materials.
Definition of iron in physiology
Iron = an essential trace mineral required mainly for oxygen transport and cellular oxidation reactions (because it forms part of haemoglobin/myoglobin and several oxidase enzymes).
Total body iron content
RBC iron (in Hb) ≈ ~3 g (total quantity present in all red cells).
Rest of the body ≈ ~1–3 g (tissue iron pool).
Whole blood contains ~45–50 mg iron/100 mL.
Distribution in body
Haemoglobin (major share in blood)
Hb contains ~92–98% of total blood iron.
Myoglobin
Iron is present as iron-porphyrin in myoglobin (muscle oxygen store).
Enzymes
Iron-containing enzymes include catalase, cytochrome, peroxidase (tissue oxidation).
Storage form
Stored mainly as ferritin (water-soluble) and haemosiderin (granular, insoluble).
Storage sites: reticuloendothelial system—especially liver, spleen, bone marrow.
Transport/other forms
Iron also exists in functional pools like transferrin, ferritin, haemosiderin.
Importance in oxygen transport
Hb formation is the primary function of iron.
Oxygen carriage: 1 g Hb carries ~1.34 mL O₂ when fully saturated.
Myoglobin supplies O₂ to muscle and acts as an oxygen store.
Clinical relevance
Body iron is controlled mainly by regulation of absorption (not excretion); excretion is only in traces (urine/bile/faeces).
Iron loss states: pregnancy, labour, menstrual blood loss → can predispose to iron deficiency.
If iron loss exceeds absorption → Hb falls → anaemia develops.
4
Site of absorption
Mainly in the duodenum and upper jejunum.
Enterocytes of proximal small intestine are specially adapted for iron uptake.
Forms of dietary iron
Heme iron
Present in animal sources (meat, liver, fish).
Absorbed intact via heme carrier protein.
Better absorbed (less affected by dietary factors).
Non-heme iron
Present in plant sources (green leafy vegetables, cereals, pulses).
Usually in ferric (Fe³⁺) form.
Must be reduced to ferrous (Fe²⁺) form for absorption.
Absorption is less efficient and more variable.
Factors affecting absorption
Increased by:
Iron deficiency state
Increased erythropoiesis
Pregnancy
Vitamin C
Decreased by:
Phytates (cereals)
Oxalates
Tannins (tea)
Excess calcium
Role of gastric acid
Converts ferric (Fe³⁺) → ferrous (Fe²⁺) form.
Maintains iron in soluble form.
Achlorhydria → ↓ iron absorption.
Role of vitamin C
Reduces Fe³⁺ → Fe²⁺.
Forms soluble complexes with iron.
Enhances non-heme iron absorption.
Regulation by body iron stores
Low iron stores → ↑ absorption.
High iron stores → ↓ absorption.
Regulation occurs mainly at the level of intestinal mucosa.
Hepcidin regulation (concept)
Hepcidin = hormone produced by liver.
High hepcidin:
Blocks ferroportin (iron exporter) in enterocytes and macrophages.
↓ iron release into plasma.
Low hepcidin:
↑ ferroportin activity.
↑ iron absorption and release.
Acts as the master regulator of iron metabolism.
Transport in plasma
Iron transported in plasma bound to transferrin.
Each transferrin molecule binds two Fe³⁺ ions.
Transferrin delivers iron mainly to:
Bone marrow (for Hb synthesis)
Liver (for storage)
Other tissues (for enzymes)


4
Reduction of ferric (Fe³⁺) to ferrous (Fe²⁺) form
Dietary non-heme iron is mainly in ferric (Fe³⁺) form.
At the brush border of enterocytes, Fe³⁺ is reduced to ferrous (Fe²⁺) form.
This reduction is essential because only Fe²⁺ can be transported into the cell.
Transport across enterocyte membrane
Fe²⁺ crosses the apical membrane of enterocyte.
Occurs mainly in the duodenum.
Role of divalent metal transporter (DMT1 concept)
DMT1 (Divalent Metal Transporter-1) transports Fe²⁺ into enterocyte.
It is a proton-coupled transporter.
Also transports other divalent metals (e.g., Mn²⁺).
Storage as ferritin in enterocytes
Inside enterocyte, iron may:
Bind to ferritin for temporary storage.
If not required by body → stored iron is lost when enterocytes are shed.
This is called the mucosal block theory (physiological control mechanism).
Transfer to plasma via ferroportin
If body requires iron:
Fe²⁺ exits enterocyte through ferroportin (basolateral membrane iron exporter).
Ferroportin is the only known iron export protein.
Oxidation to ferric form
During transfer to plasma:
Fe²⁺ is oxidized back to Fe³⁺.
This step is necessary for binding to transferrin.
Regulation mechanism
Controlled mainly by body iron stores and hepcidin.
High hepcidin:
Degrades ferroportin.
↓ iron absorption.
Low hepcidin:
↑ ferroportin activity.
↑ iron absorption.
Thus, regulation occurs at the level of intestinal absorption, not excretion.
Clinical relevance
Achlorhydria → ↓ Fe³⁺ reduction → ↓ absorption.
Chronic blood loss → ↑ iron absorption (compensatory).
Inflammation → ↑ hepcidin → anemia of chronic disease.
Excess absorption → risk of iron overload states.


4
Transport protein – Transferrin
Iron circulates in plasma bound to transferrin (a β-globulin).
Synthesized in the liver.
Each transferrin molecule binds two ferric (Fe³⁺) ions.
Prevents free iron toxicity and delivers iron to tissues (especially bone marrow).
Iron-binding capacity
Refers to the ability of transferrin to bind iron.
Depends on the amount of transferrin available in plasma.
Serum iron
Represents the amount of iron bound to transferrin in plasma.
Normal value:
Males: ~60–170 µg/dL
Females: ~50–150 µg/dL
Shows diurnal variation (higher in morning).
Total Iron-Binding Capacity (TIBC)
Measures the maximum amount of iron that transferrin can bind.
Indirect measure of transferrin level.
Normal value: 250–400 µg/dL.
Transferrin saturation
Percentage of transferrin binding sites occupied by iron.
Formula:
Transferrin saturation (%) = (Serum iron / TIBC) × 100
Normal value: 20–45%.
Normal values (summary)
Serum iron: ~60–170 µg/dL
TIBC: ~250–400 µg/dL
Transferrin saturation: 20–45%
Clinical significance
Iron deficiency anemia
↓ Serum iron
↑ TIBC
↓ Transferrin saturation
Anemia of chronic disease
↓ Serum iron
↓ or normal TIBC
↓ Transferrin saturation
Iron overload (e.g., hemochromatosis)
↑ Serum iron
↓ TIBC
↑ Transferrin saturation

4
Ferritin
Main storage form of iron.
Water-soluble protein complex.
Stores iron in ferric (Fe³⁺) form.
Present in cytoplasm of cells.
Serum ferritin reflects body iron stores.
Hemosiderin
Insoluble, granular storage form.
Formed when iron stores are excessive.
Seen as coarse golden-brown granules on microscopy.
Less readily available for mobilization than ferritin.
Sites of storage
Liver
Major storage organ.
Iron stored in hepatocytes and Kupffer cells.
Spleen
Stores iron from breakdown of old RBCs.
Bone marrow
Iron available for hemoglobin synthesis.
Regulation of storage
Controlled mainly by body iron requirements and hepcidin.
High iron levels → increased storage.
Low iron levels → decreased storage.
Balance maintained between absorption, utilization, and storage.
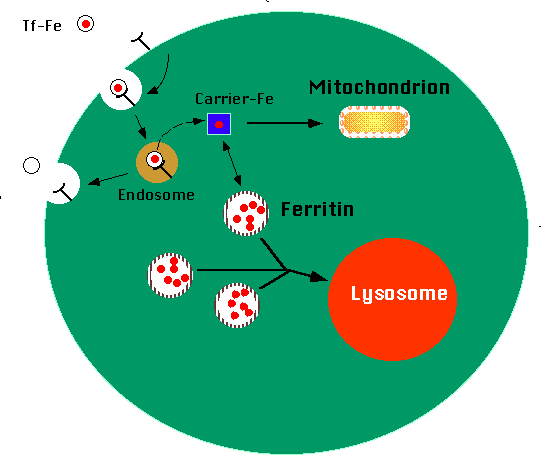
Mobilization of stored iron
Iron released from ferritin when needed.
Exported via ferroportin.
Binds to transferrin in plasma for transport to bone marrow.
Enhanced during anemia and increased erythropoiesis.
Clinical importance
Serum ferritin = best indicator of total body iron stores.
↓ Ferritin → iron deficiency.
↑ Ferritin → iron overload, inflammation, liver disease.
Excess deposition → hemosiderosis, hemochromatosis.
4
Hemoglobin synthesis
Essential component of heme molecule.
Required for formation of hemoglobin (Hb) in RBCs.
Each Hb molecule contains four heme groups, each with one iron atom.
Enables reversible binding of oxygen.
Without iron → defective Hb synthesis → microcytic hypochromic anemia.
Myoglobin formation
Present in skeletal and cardiac muscle.
Acts as oxygen reservoir.
Facilitates diffusion of oxygen within muscle cells.
Important during intense muscular activity.
Enzyme function
Component of several iron-containing enzymes, such as:
Catalase
Peroxidase
Cytochromes
Participates in oxidation–reduction reactions.
Essential for detoxification of hydrogen peroxide.
Role in electron transport chain
Iron is part of cytochromes and iron–sulfur (Fe–S) proteins.
Facilitates electron transfer in mitochondria.
Critical for ATP production.
Without iron, oxidative phosphorylation is impaired.
Role in oxidative metabolism
Required for cellular respiration.
Participates in redox reactions.
Supports efficient utilization of oxygen in tissues.
Importance in cell growth and division
Required for DNA synthesis (via ribonucleotide reductase enzyme).
Essential for rapidly dividing cells (bone marrow, fetal tissues).
Deficiency leads to:
Impaired growth
Reduced cognitive development in children
Decreased immunity

4
Causes
Nutritional deficiency
Poor dietary intake.
Diet low in heme iron (vegetarian diets without adequate supplementation).
Common in children and low socioeconomic groups.
Blood loss
Chronic gastrointestinal bleeding.
Menstrual blood loss (menorrhagia).
Hookworm infestation.
Repeated blood donation.
Malabsorption
Celiac disease.
Post-gastrectomy state.
Achlorhydria (↓ gastric acid → ↓ Fe³⁺ to Fe²⁺ conversion).
Increased requirement
Pregnancy.
Lactation.
Infancy and adolescence (rapid growth phase).
Pathophysiology
↓ Iron stores → ↓ serum ferritin.
↓ Iron availability for hemoglobin synthesis.
Impaired heme formation.
Reduced hemoglobin production in developing RBCs.
Result: smaller, paler red cells.
Microcytic hypochromic anemia
Microcytic → decreased MCV.
Hypochromic → decreased MCH/MCHC.
Peripheral smear shows:
Small RBCs.
Increased central pallor.
Anisopoikilocytosis (variation in size and shape).
Clinical features
Pallor (skin, conjunctiva).
Fatigue and weakness.
Dyspnea on exertion.
Palpitations.
Koilonychia (spoon-shaped nails).
Angular cheilitis.
Glossitis.
Pica (craving for non-food items).
Laboratory findings
↓ Hemoglobin.
↓ Serum iron.
↓ Serum ferritin (early marker).
↑ TIBC.
↓ Transferrin saturation.
↓ MCV, ↓ MCH.
Prevention principles
Iron-rich diet (green leafy vegetables, meat, liver).
Iron supplementation in:
Pregnancy.
Infants and adolescents.
Deworming in endemic areas.
Early detection and treatment of chronic blood loss.
Get the full PDF version of this chapter.